(wait until the page is loaded completely)
CleanImplant "Trusted Quality" Mark
Implant systems that comply with the CleanImplant Quality Mark Criteria according to the consensus recommendation of the Scientific Advisory Board are listed below. This guideline for clean implants was released September 2017 and has yet been established in many implant manufactoring sites as de-facto standard for the cleanliness of implantable devices.
Click on the Quality Mark image to download the consensus paper (4 MByte PDF-file).
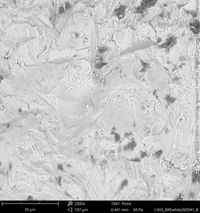
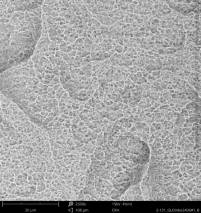
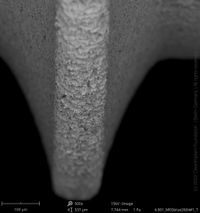
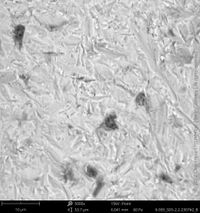
For the comprehensive quality assessment five implant samples of each implant type have been analyzed by means of SEM and elemental analysis. The certification "Trusted Quality" is valid for two years before the CleanImplant re-evaluation process has to start. Hence, the list of implant systems (in alphabetic order) carrying the "Trusted Quality Mark" is continually updated and extended.
The implant system of your choice is not listed below ? Please do not hesitate and send us a mail !
"Trusted Quality" Implant Types 2023 (in alphabetic order)
ASTRA TECH EV - Dentsply Sirona
Click on the images to enlarge
Biotech Dental – Kontact S
Click on the images to enlarge
bredent medical – whiteSKY
Click on the images to enlarge.
BTI Biotechnology Institute - UnicCa
Click on the images to enlarge.
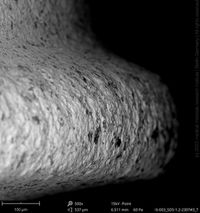
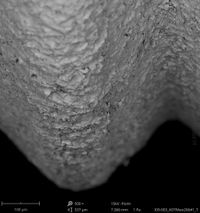
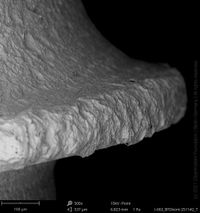
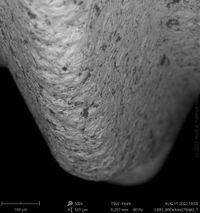
Champions Implants - R(E)volution
Click on the images to enlarge
Dentis - s-Clean SQ-SL
Click on the images to enlarge
Dentium – SuperLine
Click on the images to enlarge
Global D – In-Kone
Click on the images to enlarge.
Medentis Medical – ICX-Premium - Re-evaluation currently in peer-review
Click on the images to enlarge.
Click on the images to enlarge.
Ritter Implants - Spiral SB/LA
Click on the images to enlarge.
Southern Implants - INVERTA
Click on the images to enlarge.
Swiss Dental Solutions - SDS2.2
Click on the images to enlarge.
Swiss Dental Solutions - SDS1.2
Click on the images to enlarge.
NOTE:




















Evolution_22-00010-002_SEM-Mapping.jpg/picture-200?_=183760c2538)
Evolution_22-00010-002_Thread_500x.jpg/picture-200?_=183760c30f0)
Evolution%20_2022-2024.jpg/picture-200?_=183760c1d68)
Evolution_22-00010-002_500x.jpg/picture-200?_=183760c2920)
Evolution_22-00010-002_2500x.jpg/picture-200?_=183760c2920)
Evolution_22-00010-002_5000x.jpg/picture-200?_=183760c2d08)




















































.jpg/picture-200?_=18376110350)