Quality Seal for Dental Implants
More safety for patients and practitioners
Dr Michael Norton, former president of the Academy of Osseointegration, summed up a problem of the implant market, stating “Dentists have to rely on the word of manufacturers and the FDA or CE marks to feel sure that the implants they are using are being manufactured to a standard one would expect of an implantable dental device. Sadly, this is often not the case.”
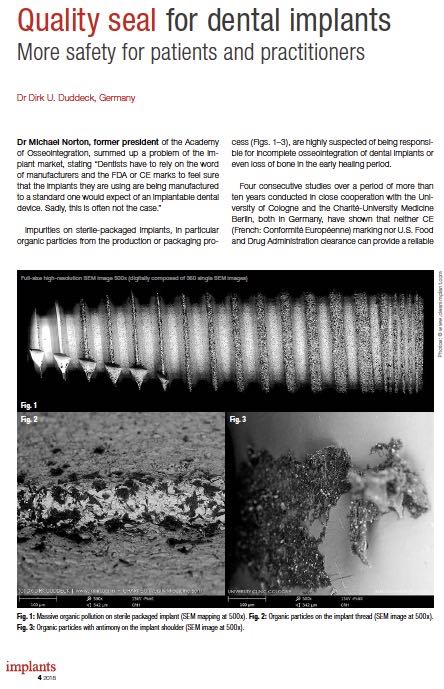
Impurities on sterile-packaged implants, in particular organic particles from the production or packaging process (Figs. 1–3), are highly suspected of being responsible for incomplete osseointegration of dental implants or even loss of bone in the early healing period.
Four consecutive studies over a period of more than ten years conducted in close cooperation with the University of Cologne and the Charité-University MedicineBerlin, both in Germany, have shown that neither CE (French: Conformité Européenne) marking nor U.S. Food and Drug Administration clearance can provide a reliable
Article by Dr. Dirk U. Duddeck, published in: implants 4-2018
Read the Full article here